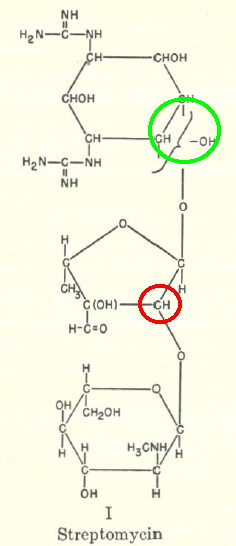
Sometimes, connections between different areas of chemistry just pop out (without the help of semantic web tools, this is called serendipity). So here, I will try to join up some threads which emerge from previous posts. I had noted that antiaromaticity in cyclopropenium anion is lessened by the system adopting gross geometric distortions, which take the anionic lone pair out of conjugation from the ring.