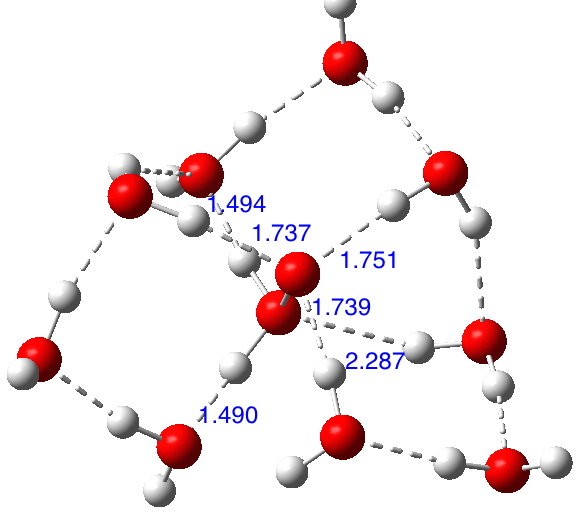
If H3N+-O– is viable compared with its tautomer H2N-OH when carrying water bridges, then why not try H2O+-O– vs HO-OH? https://orcid.org/0000-0002-8635-8390

If H3N+-O– is viable compared with its tautomer H2N-OH when carrying water bridges, then why not try H2O+-O– vs HO-OH? https://orcid.org/0000-0002-8635-8390
In the previous post I described how hydronium hydroxide or H3O+…HO–, an intermolecular tautomer of water, has recently been observed captured inside an organic cage[cite]10.1002/chem.201406383[/cite] and how the free-standing species in water can be captured computationally with the help of solvating water bridges.
Ammonium hydroxide (NH4+…OH–) can be characterised quantum mechanically when stabilised by water bridges connecting the ion-pairs.
Publishing embargoes seem a relatively new phenomenon, probably starting in areas of science when the data produced for a scientific article was considered more valuable than the narrative of that article. However, the concept of the embargo seems to be spreading to cover other aspects of publishing, and I came across one recently which appears to take such embargoes into new and uncharted territory.
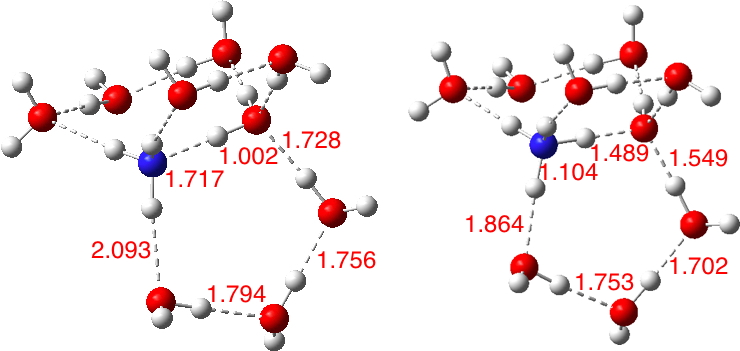
Previously, I looked at models of how ammonia could be protonated by water to form ammonium hydroxide.
A celebration of the life and work of the great chemist Paul von R. Schleyer was held this week in Erlangen, Germany. There were many fantastic talks given by some great chemists describing fascinating chemistry. Here I highlight the presentation given by Andy Streitwieser on the topic of organolithium chemistry, also a great interest of Schleyer's over the years.
Augmented reality, a superset if you like of virtual reality (VR), has really been hitting the headlines recently. Like 3D TV, its been a long time coming! Since ~1994 or earlier, there have been explorations of how molecular models can be transferred from actual reality to virtual reality using conventional computers (as opposed to highly specialised ones). It was around then that a combination of software (Rasmol) and
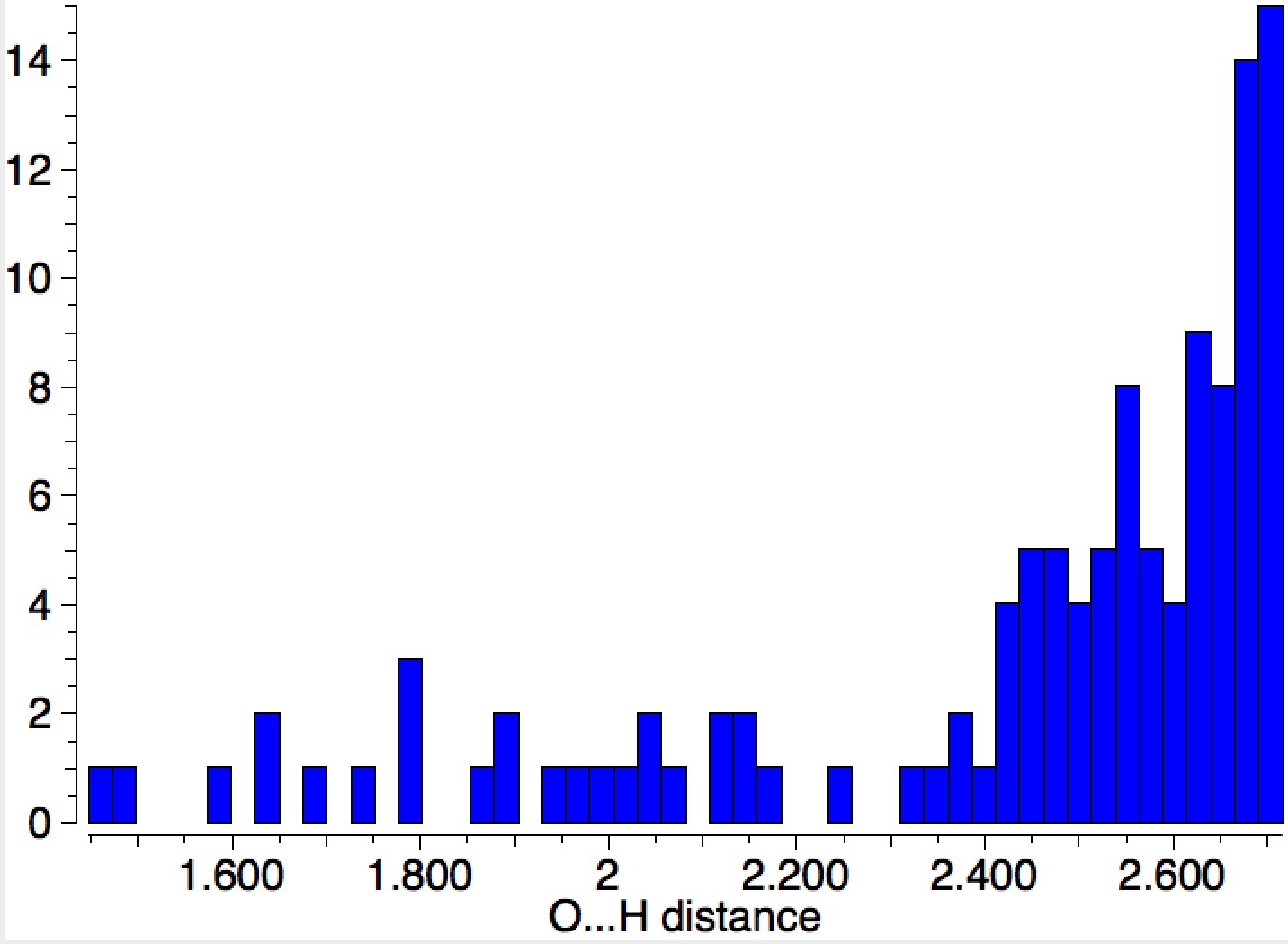
This is a corollary to the previous post ‡ exploring how many molecules are needed to ionise HCl. Here I am asking how many water molecules are required to form the ionic ammonium hydroxide from ammonia and water.
At the ACS conference, I have attended many talks these last four days, but one made some “connections” which intrigued me. I tell its story (or a part of it) here. But to start, try the following experiment. Find a Word document of .docx type on your hard drive Remove the .docx suffix and replace it with a .zip suffix. Expand as if it is an archive (it is!). A folder is created and this itself contains four further folders.
The upcoming ACS national meeting in San Diego has a CINF (chemical information division) session entitled "Global initiatives in research data management and discovery". I have highlighted here just one slide from my contribution to this session, which addresses the discovery aspect of the session.