
I previously blogged about anomeric effects involving π electrons as donors, and my post on the conformation of 1,2-difluorethane turned out one of the most popular.

I previously blogged about anomeric effects involving π electrons as donors, and my post on the conformation of 1,2-difluorethane turned out one of the most popular.
One frequently has to confront the question: will a hydrogen bond form between a suitable donor (lone pair or π) and an acceptor? One of the factors to be taken into consideration for hydrogen bonds which are part of a cycle is the ring size. Here I explore one way of quantifying the effect for the series below, n=1-5 (4-8 membered rings). I will use the NBO approach.
I return to this reaction one more time. Trying to explain why it is enantioselective for the epoxide product poses peculiar difficulties. Most of the substituents can adopt one of several conformations, and some exploration of this conformational space is needed. Amongst the conformational possibilities are the two rotations shown below.
tpap[cite]10.1055%2Fs-1994-25538[/cite], as it is affectionately known, is a ruthenium-based oxidant of primary alcohols to aldehydes discovered by Griffith and Ley.
I have written earlier about dihydrocostunolide, and how in 1963 Corey missed spotting the electronic origins of a key step in its synthesis.[cite]10.1021/ja00952a037[/cite]. A nice juxtaposition to this failed opportunity relates to Woodward’s project at around the same time to synthesize vitamin B12. The step in the synthesis that caused him to ponder is shown below.
The Sharpless epoxidation of an allylic alcohol had a big impact on synthetic chemistry when it was introduced in the 1980s, and led the way for the discovery (design?) of many new asymmetric catalytic systems. Each achieves its chiral magic by control of the geometry at the transition state for the reaction, and the stabilizations (or destabilizations) that occur at that geometry.
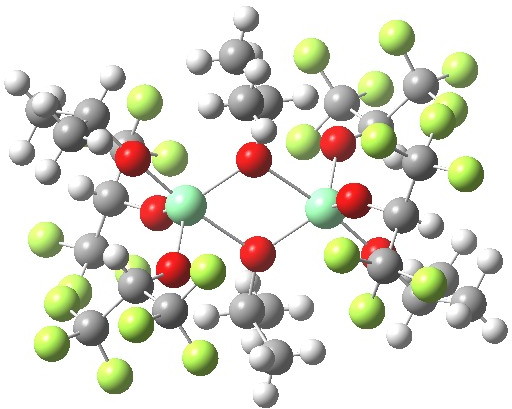
Part one on this topic showed how a quantum mechanical model employing just one titanium centre was not successful in predicting the stereochemical outcome of the Sharpless asymmetric epoxidation.
Sharpless epoxidation converts a prochiral allylic alcohol into the corresponding chiral epoxide with > 90% enantiomeric excess[cite]10.1021/jo00369a032[/cite],[cite]10.1021/jo00360a058[/cite]. Here is the first step in trying to explain how this magic is achieved.
I was intrigued by one aspect of the calculated transition state for di-imide reduction of an alkene; the calculated NMR shieldings indicated an diatropic ring current at the centre of the ring, but very deshielded shifts for the hydrogen atoms being transferred. This indicated, like most thermal pericyclic reactions, an aromatic transition state. Well, one game one can play with this sort of reaction is to add a double bond.
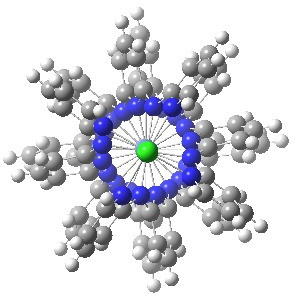
More than 60 million molecules are known, and many are fascinating. But beauty is in the eye of the beholder. Thus it was that I came across the attached molecule[cite]10.1021/ja982065w[/cite]. It struck me immediately as, well, beautiful! GOCTOH. Click for 3D. This is one that comes to life in 3D and I strongly urge you to inspect it as such by clicking on the above. Why is it so interesting?