This is another of those posts that has morphed from an earlier one noting the death of the great chemist George Olah.

This is another of those posts that has morphed from an earlier one noting the death of the great chemist George Olah.
George Olah passed away on March 8th. He was part of the generation of scientists in the post-war 1950s who had access to chemical instrumentation that truly revolutionised chemistry.
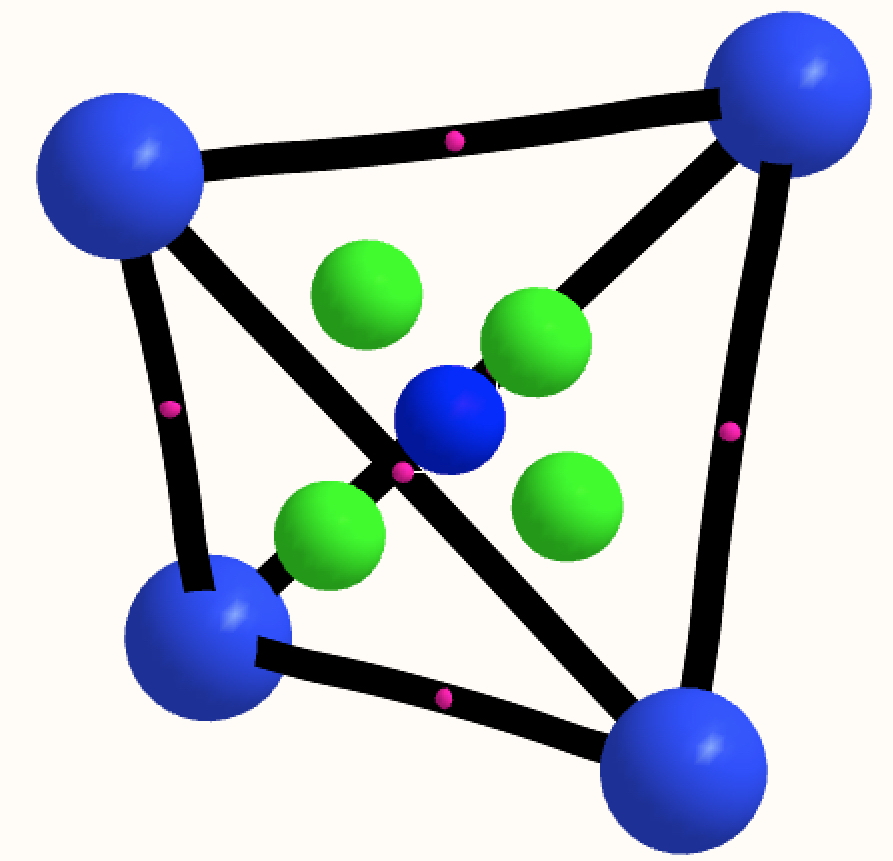
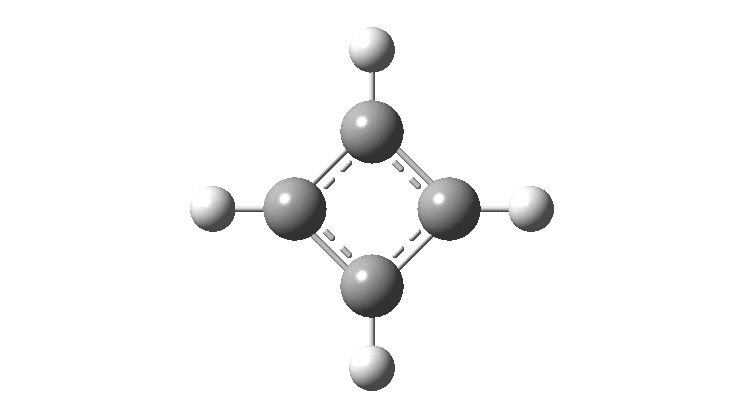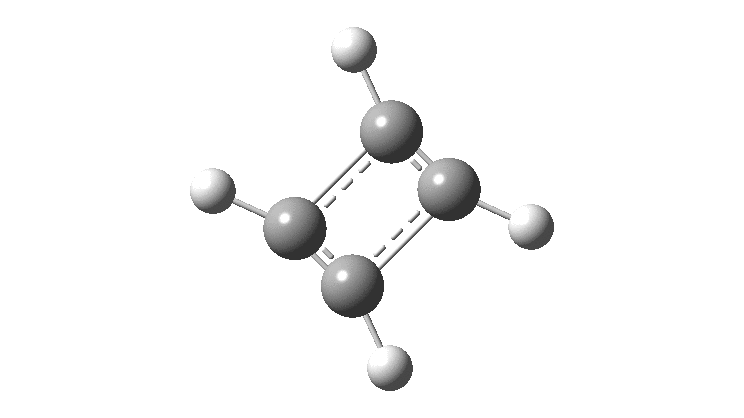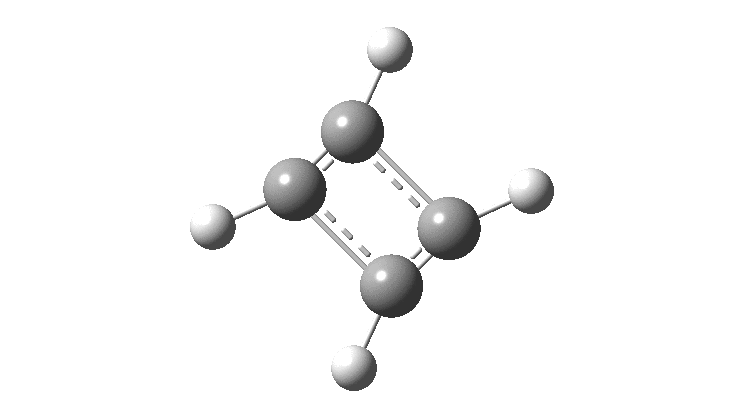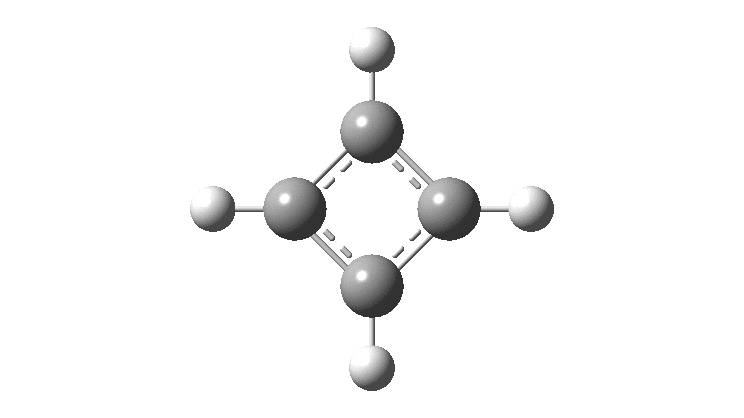
The thread thus far. The post about Na 2 He introduced the electride anionic counter-ion to Na + as corresponding topologically to a rare feature known as a non-nuclear attractor. This prompted speculation about other systems with such a feature, and the focus shifted to a tetrahedral arrangement of four hydrogen atoms as a dication, sharing a total of two valence electrons. The story now continues here.
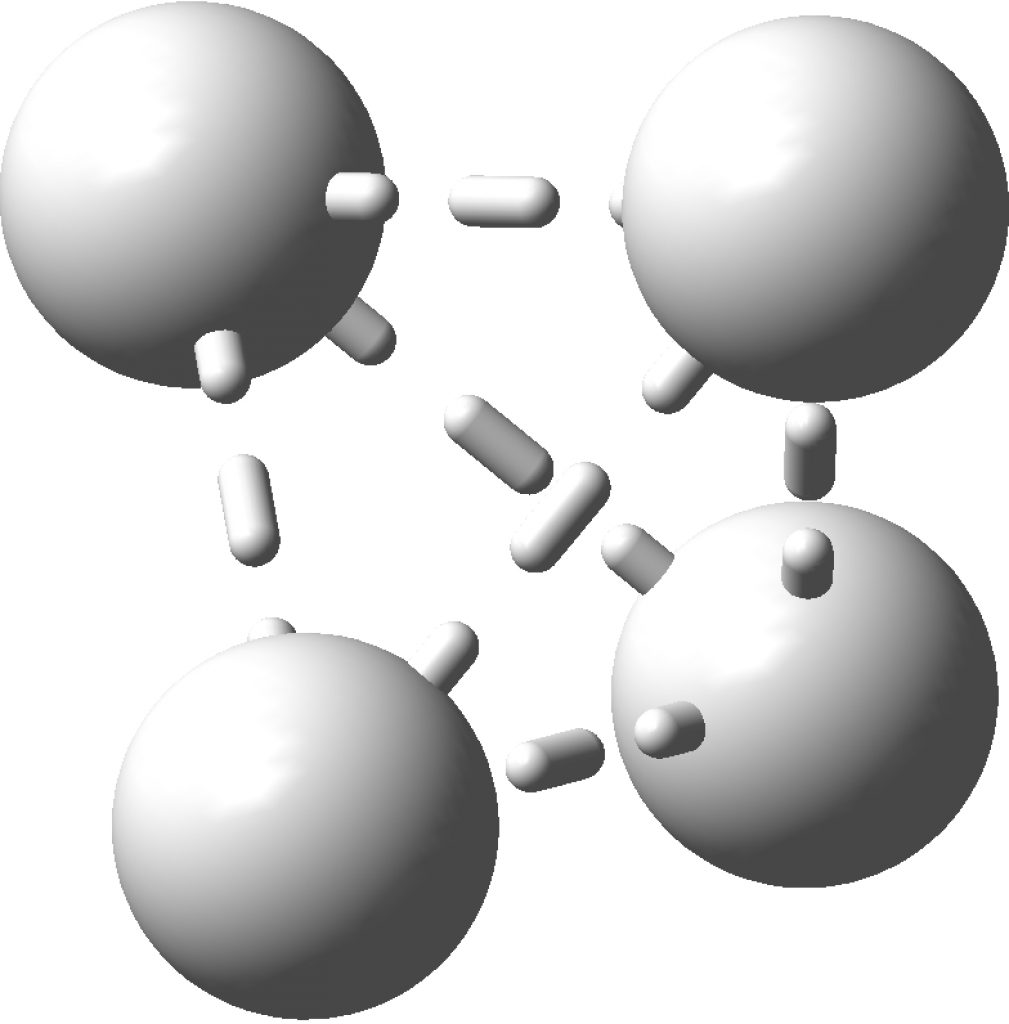
This post arose from a comment attached to the post on Na 2 He and relating to peculiar and rare topological features of the electron density in molecules called non-nuclear attractors. This set me thinking about other molecules that might exhibit this and one of these is shown below.

I analysed the bonding in chlorine trifluoride a few years back in terms of VSEPR theory. I noticed that several searches on this topic which led people to this post also included a query about the differences between it and the bromine analogue. For those who posed this question, here is an equivalent analysis.

On February 6th I was alerted to this intriguing article[cite]10.1038/nchem.2716[/cite] by a phone call, made 55 minutes before the article embargo was due to be released. Gizmodo wanted to know if I could provide an (almost) † instant ‡ quote. After a few days, this report of a stable compound of helium and sodium still seems impressive to me and I now impart a few more thoughts here.
The story so far. Inspired by the report of the most polar neutral compound yet made, I suggested some candidates based on the azulene ring system that if made might be even more polar. This then led to considering a smaller π-analogue of azulene, m-benzyne. Here I ponder how a derivative of this molecule might be made, using computational profiling as one reality check.
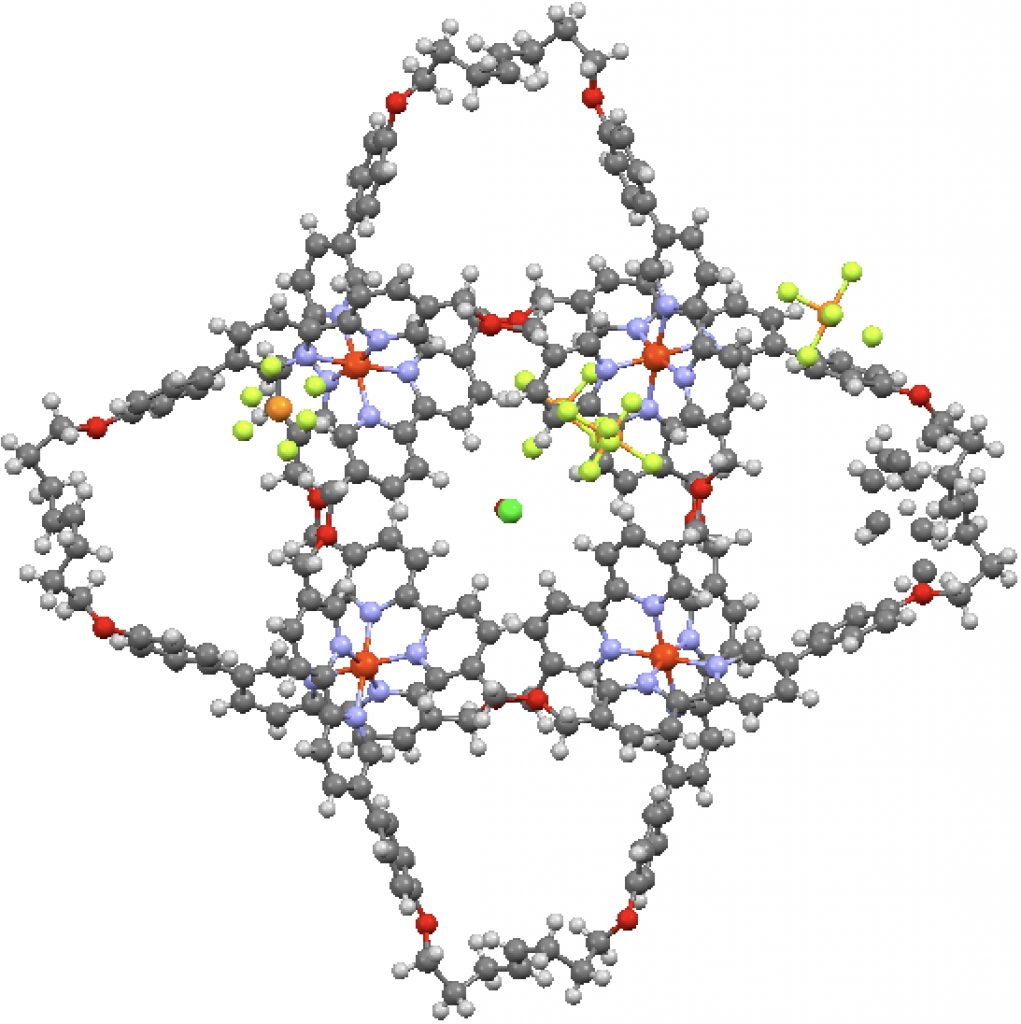
This is one of those posts of a molecule whose very structure is interesting enough to merit a picture and a 3D model. The study[cite]10.1126/science.aal1619[/cite] reports a molecular knot with the remarkable number of eight crossings.

Here is an inside peek at another one of Derek Lowe’s 250 milestones in chemistry, the polymorphism of Ritonavir .[cite]10.1023/A:1011052932607[/cite] The story in a nutshell concerns one of a pharma company’s worst nightmares;
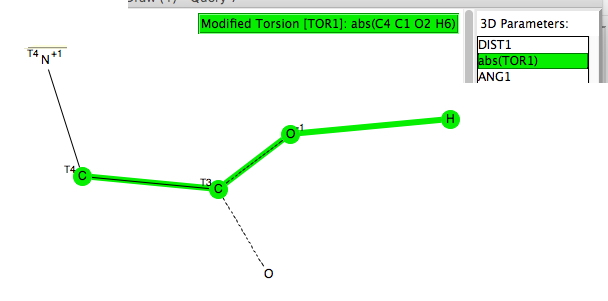
The previous posts produced discussion about the dipole moments of highly polar molecules.