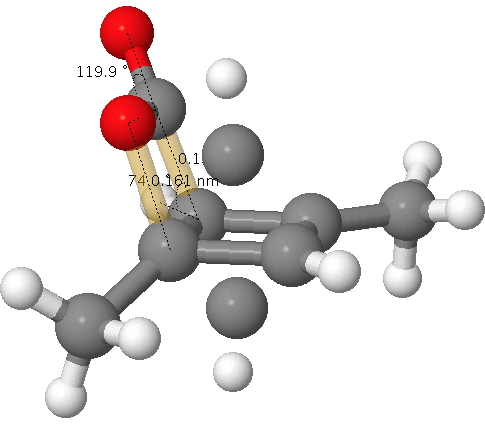
Cyclobutadiene is one of those small iconic molecules, the transience and instability of which was explained theoretically long before it was actually detected in 1965.[cite]10.1021/ja01092a049[/cite] Given that instability, I was intrigued as to how many crystal structures might have been reported for this ring system, along with the rather more stable congener cyclo-octatetraene. Here is what I found.