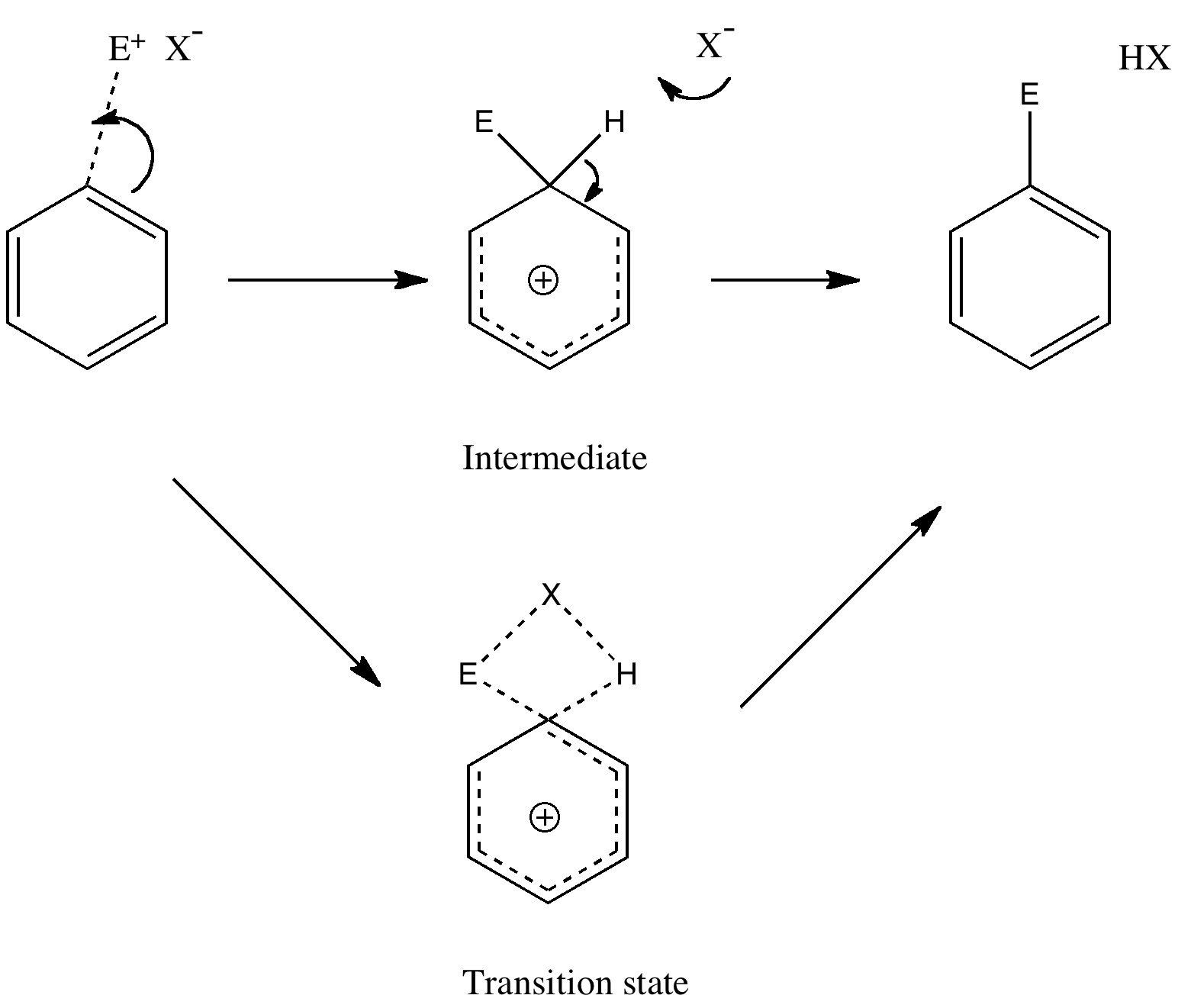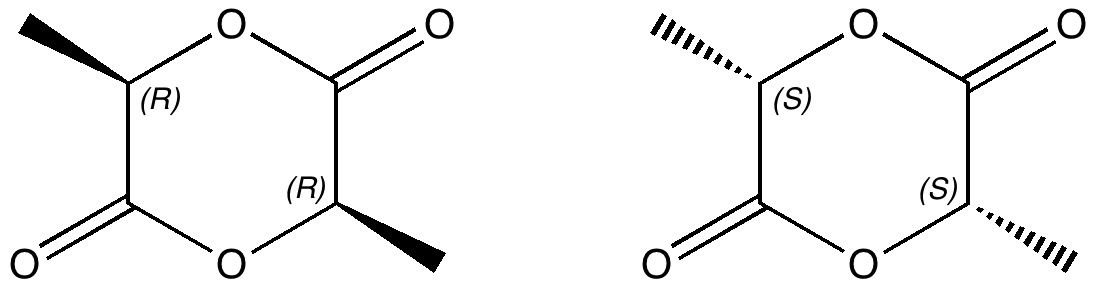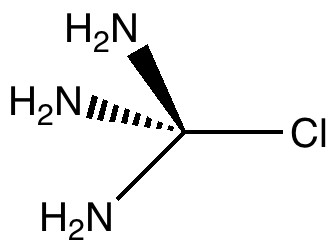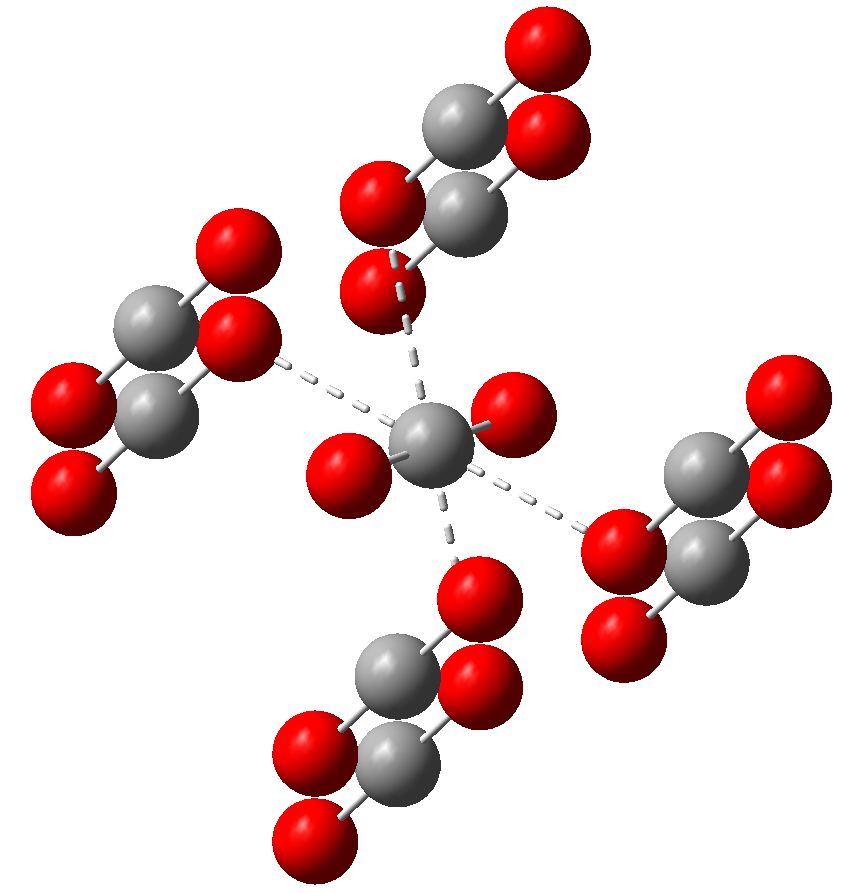
Carbon dioxide is much in the news, not least because its atmospheric concentration is on the increase. How to sequester it and save the planet is a hot topic. Here I ponder its solid state structure, as a hint to its possible reactivity, and hence perhaps for clues as to how it might be captured. The structure was determined (DOI 10.1103/PhysRevB.65.104103) as shown below. The structure of solid carbon dioxide.