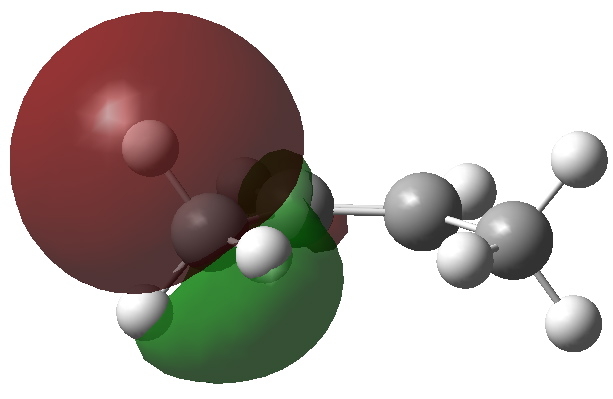
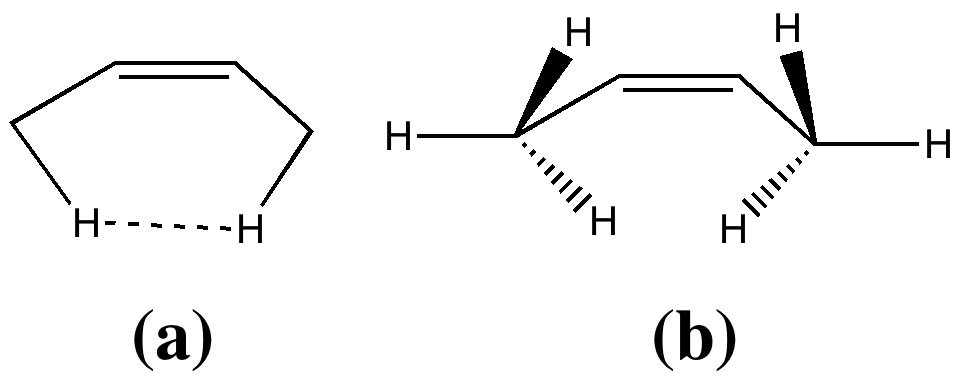
I wrote earlier about the strangely close contact between two hydrogen atoms in cis -butene. The topology of the electron density showed characteristics of a bond, but is it a consensual union?

I wrote earlier about the strangely close contact between two hydrogen atoms in cis -butene. The topology of the electron density showed characteristics of a bond, but is it a consensual union?
Steve Jobs death on October 5th 2011 was followed by a remarkable number of tributes and reflections on the impact the company he founded has had on the world. Many of these tributes summarise the effect as a visionary disruption . Here I describe from my own perspective some of the disruptions to chemistry I experienced (for another commentary, see here). Chemical diagram, circa 1983.
The properties of electrons are studied by both chemists and physicists. At the boundaries of these two disciplines, sometimes interesting differences in interpretation emerge. One of the most controversial is that due to Bader (for a recent review, see DOI: 10.1021/jp102748b) a physicist who brought the mathematical rigor of electronic topology to bear upon molecules.
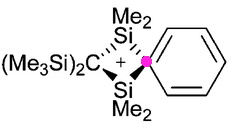
Charges in chemistry, like the grin on Lewis Carroll’s cat, can be mysterious creatures. Take for example the following structure, reported by Paul Lickiss and co-workers (DOI: 10.1039/b513203g). A silenium cation.
To (mis)quote Oscar Wilde again, ““To lose one methyl group may be regarded as a misfortune; to lose both looks like carelessness.” Here, I refer to the (past) tendency of molecular modellers to simplify molecular structures. Thus in 1977, quantum molecular modelling, even at the semi-empirical level, was beset by lost groups. One of my early efforts (DOI: 10.1021/ja00465a005) was selected for study because it had nothing left to lose;
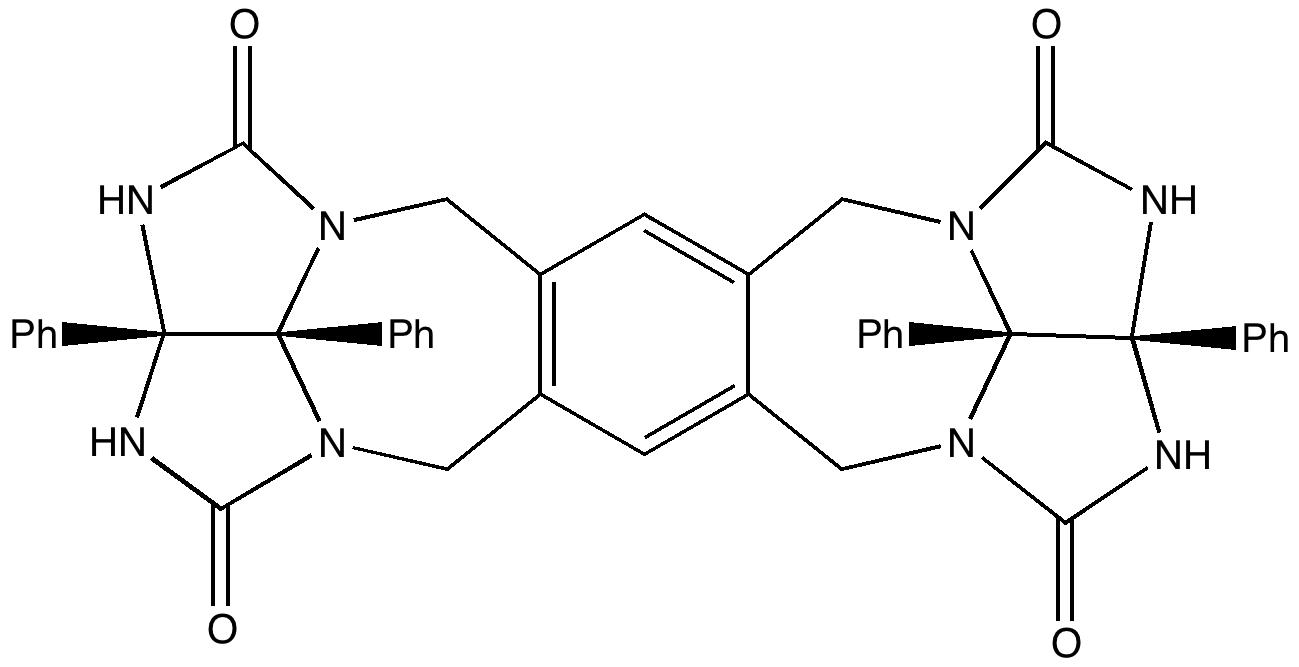
A Matryoshka doll is better known as a Russian nesting doll. They can have up to eight layers. Molecules can only emulate two layers, although see here for a good candidate for making a three-layered example (the inside layer is C 60 , which itself might encapsulate a small molecule. See also DOI: 10.1021/ja991747w). These molecular dolls can be created out of quite simple molecules.
Organic chemistry has some no-go areas, where few molecules dare venture. One of them is described by a concept known as anti-aromaticity. Whereas aromatic molecules are favoured species, their anti-equivalent is avoided. I previously illustrated this (Hückel rule) with cyclopropenium anion.
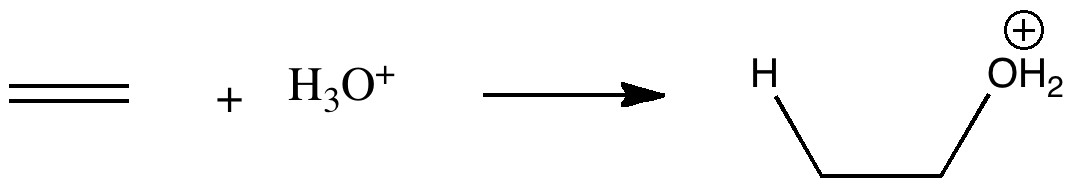
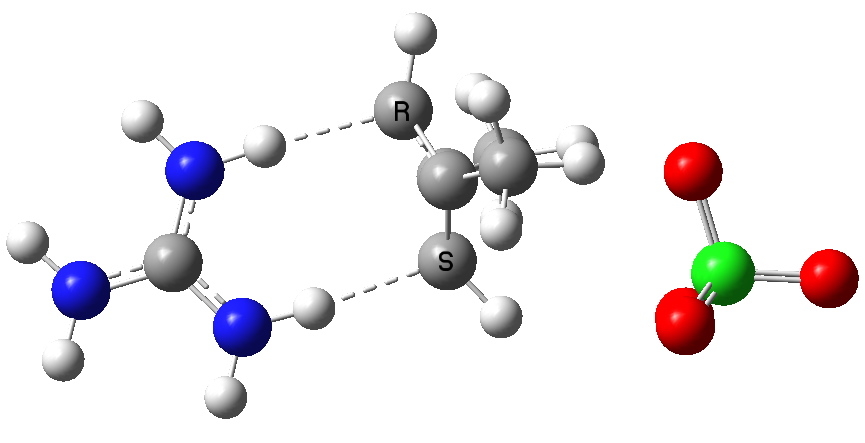
The hydration of an alkene by an acid is one of those fundamental reactions, taught early on in most chemistry courses. What can quantum mechanics teach us about the mechanism of the reaction? The hydration of ethene by a hydronium cation.
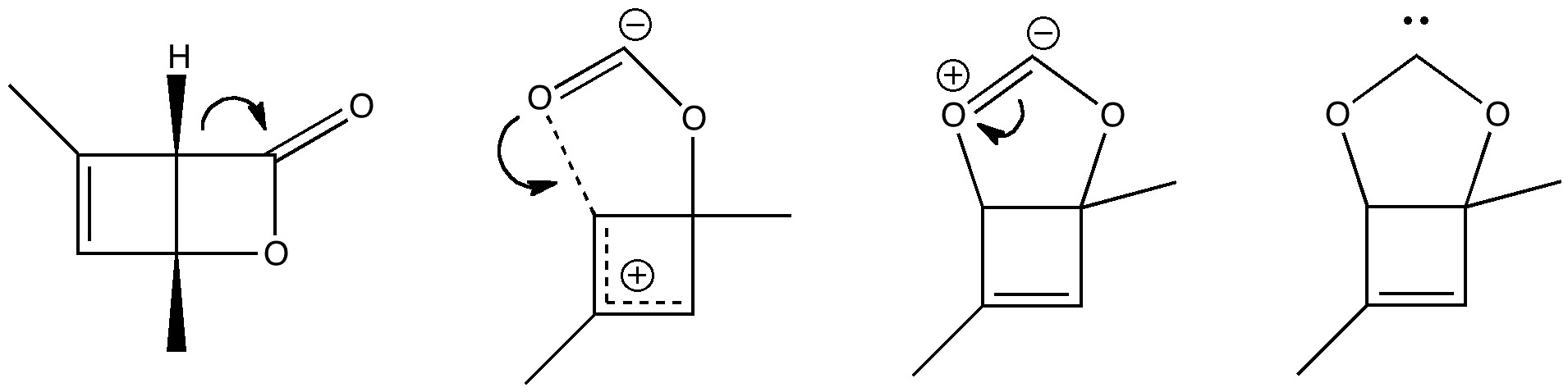
The (hopefully tongue-in-cheek) title Mindless chemistry was given to an article reporting[cite]10.1021/jp057107z[/cite] an automated stochastic search procedure for locating all possible minima with a given composition using high-level quantum mechanical calculations. “Many new structures, often with nonintuitive geometries, were found”. Well, another approach is to follow unexpected hunches.
I have mentioned Lewis a number of times in these posts; his suggestion of the shared electron covalent bond still underpins much chemical thinking.