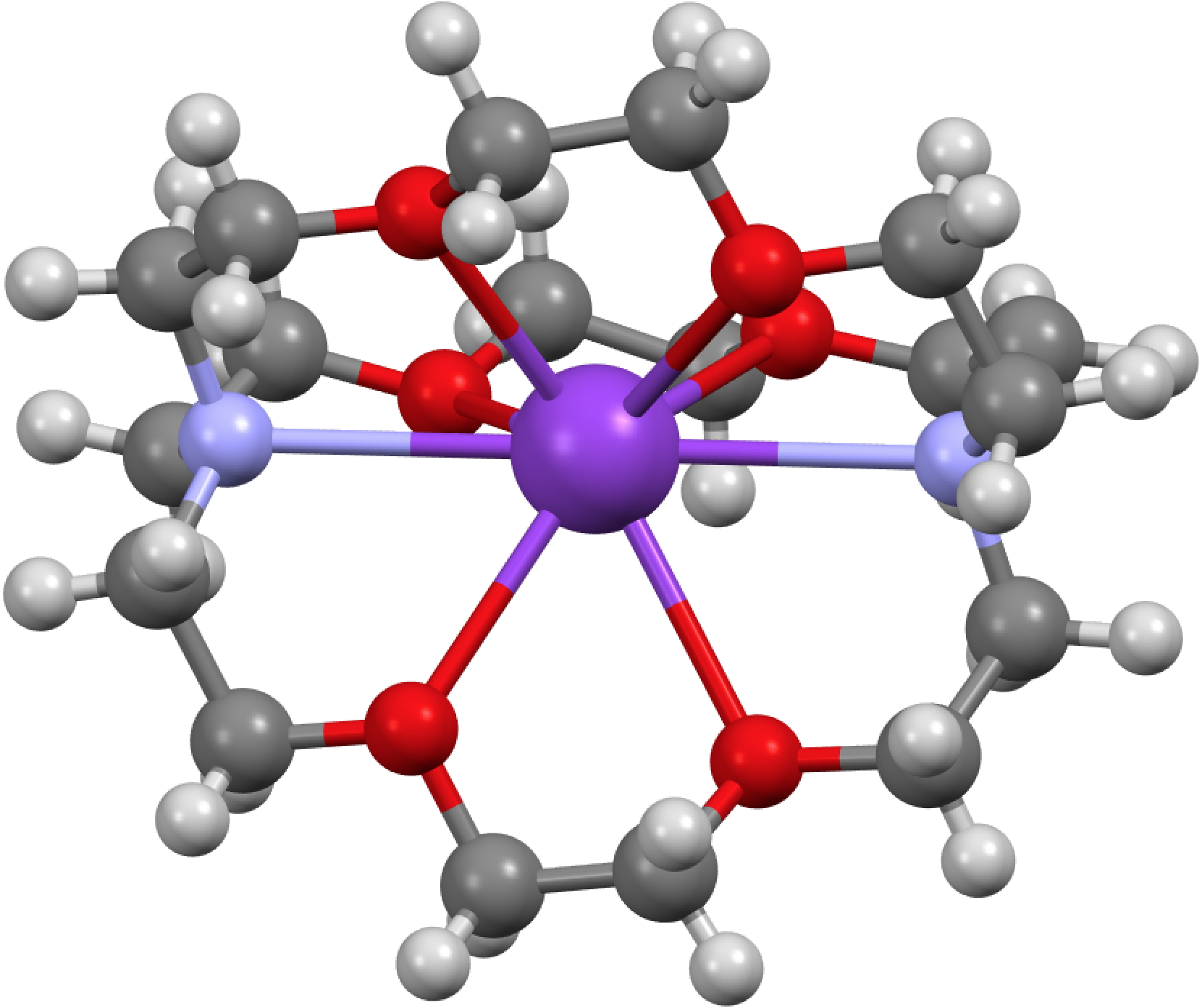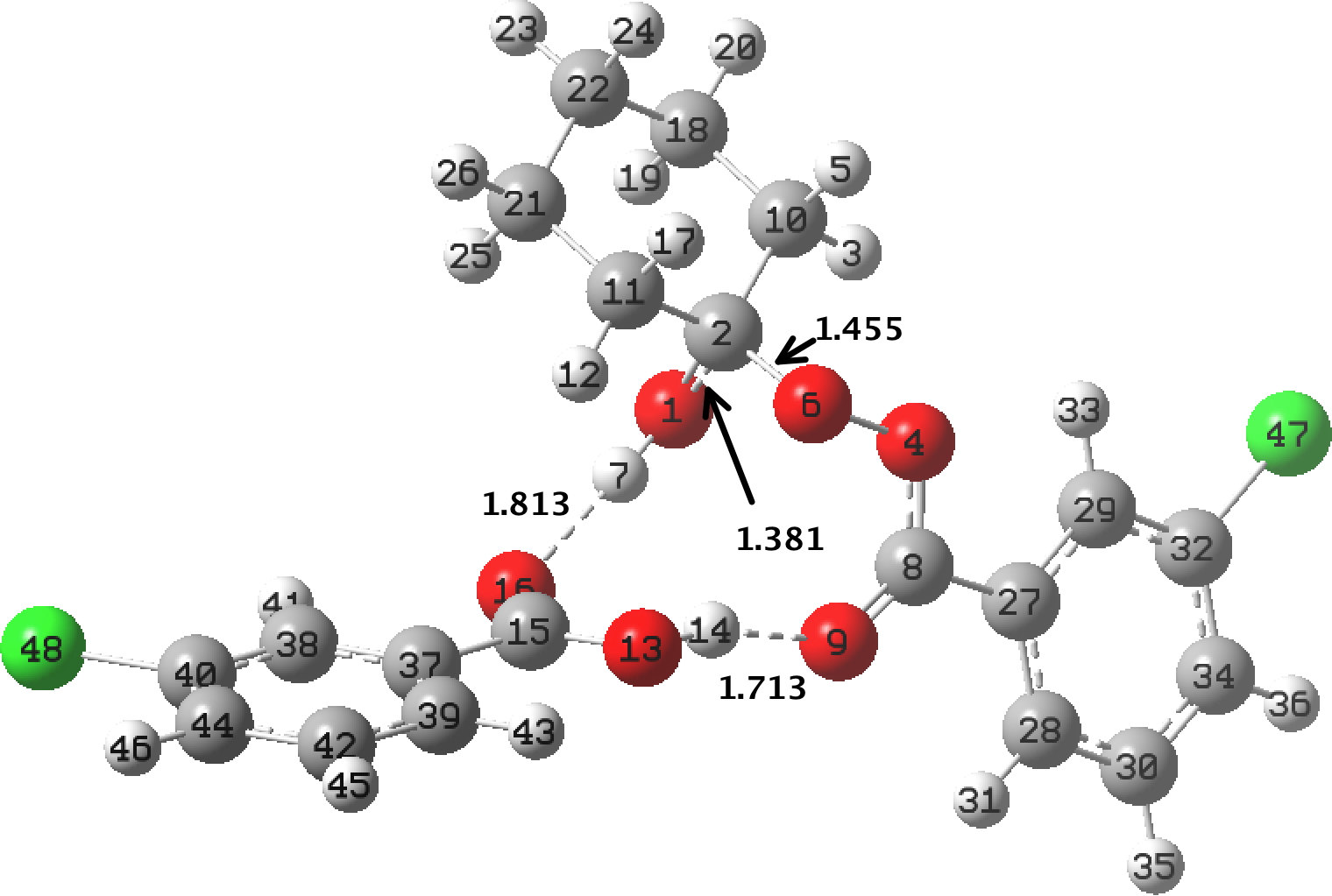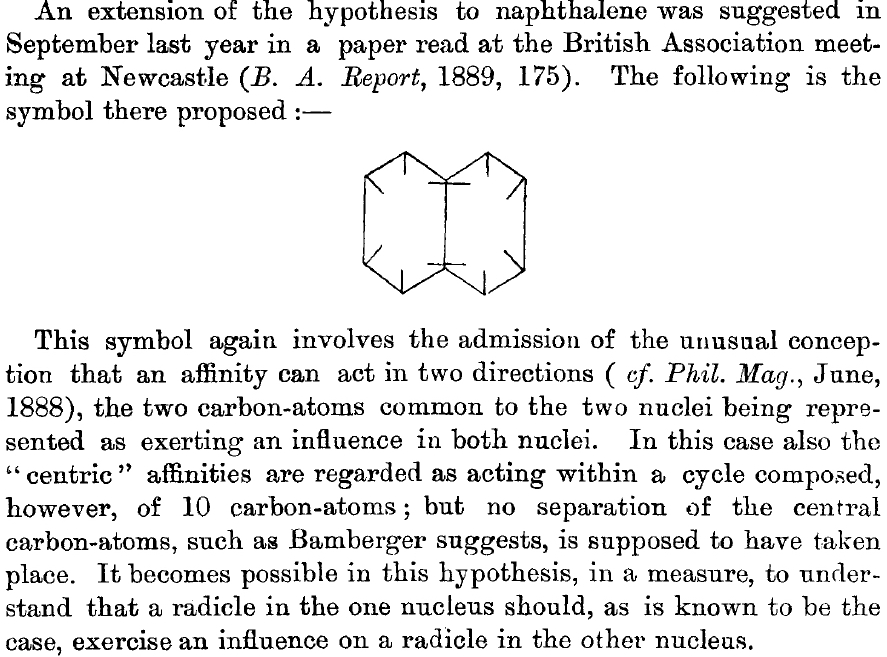
This is a little historical essay into the electronic structure of naphthalene, presented as key dates (and also collects comments made which were appended to other posts). 1890[cite]10.1039/pl8900600095[/cite]: Henry Armstrong presents the following structure of naphthalene. Three words need translation into modern usage. Where he uses the word nuclei the closest translation now might be rings .