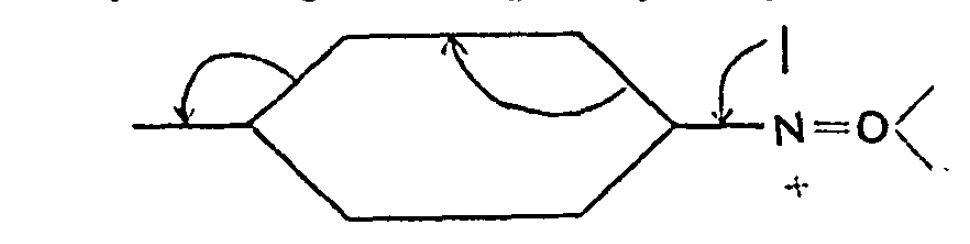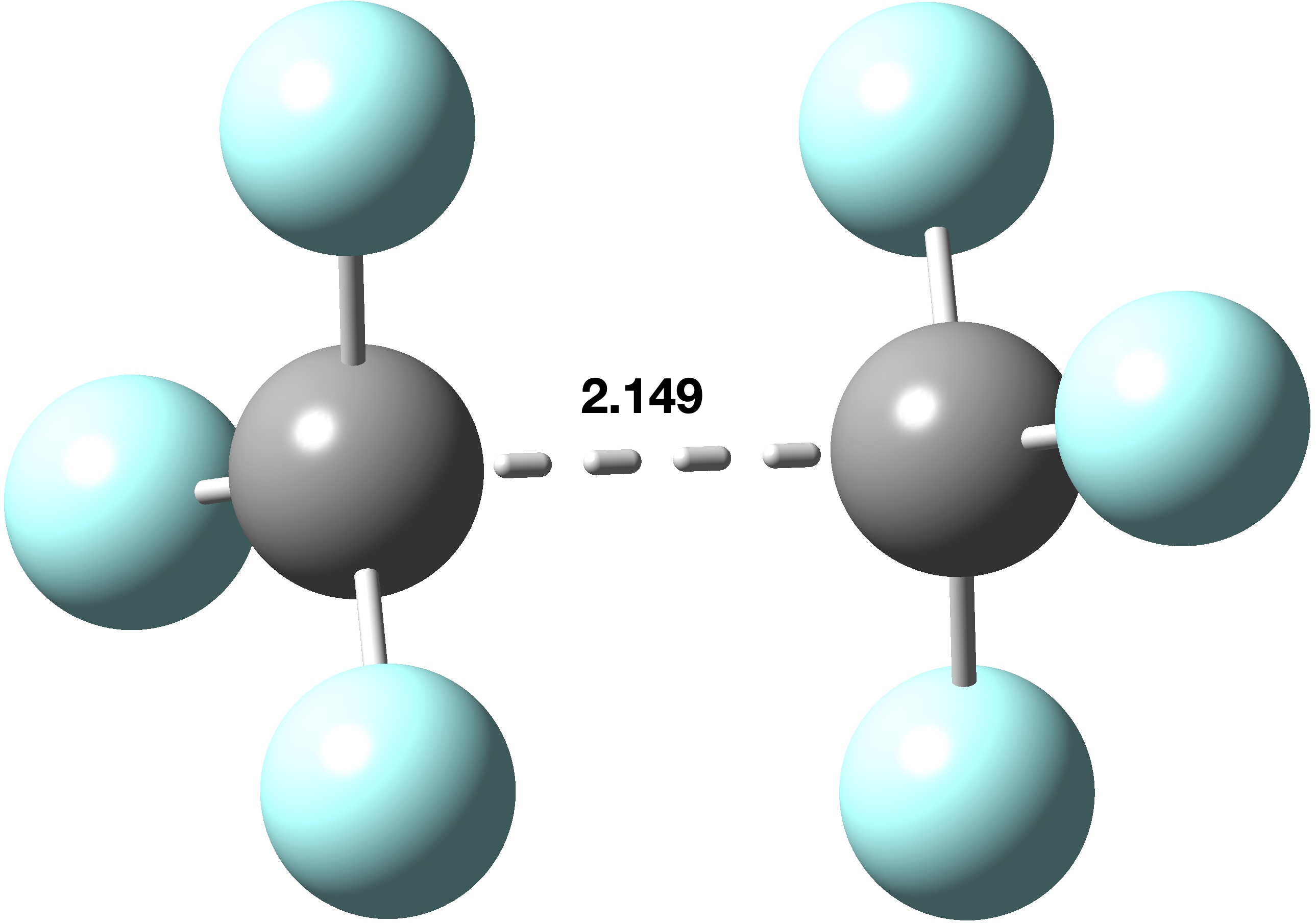
In the previous post, I looked[cite]10.59350/xp5a3-zsa24[/cite] at the recently reported[cite]10.1021/ja02261a002[/cite] hexa-arylethane containing a carbon-carbon one-electron bond, its structure having been determined by x-ray diffraction (XRD). The measured C-C bond length was ~2.9aÅ and my conclusion was that the C…C region represented more of a weak “interaction” than of a bond as such. How about a much simpler system,